Sodium: The Explosive Start of the Third Period. When One Extra Nucleon Changes Everything. Part 14
“One stone is enough to break a dam.”
— Folk wisdom
In the previous article we admired Neon — an architectural masterpiece of symmetry. Five alpha particles, perfectly packed into a “cross,” created an airtight fortress that no chemical reagent could penetrate.
But what happens if we try to add one more brick to this perfect form?
Welcome to the third period. Sodium takes the stage.
📐 Engineering Analysis of the Nucleus
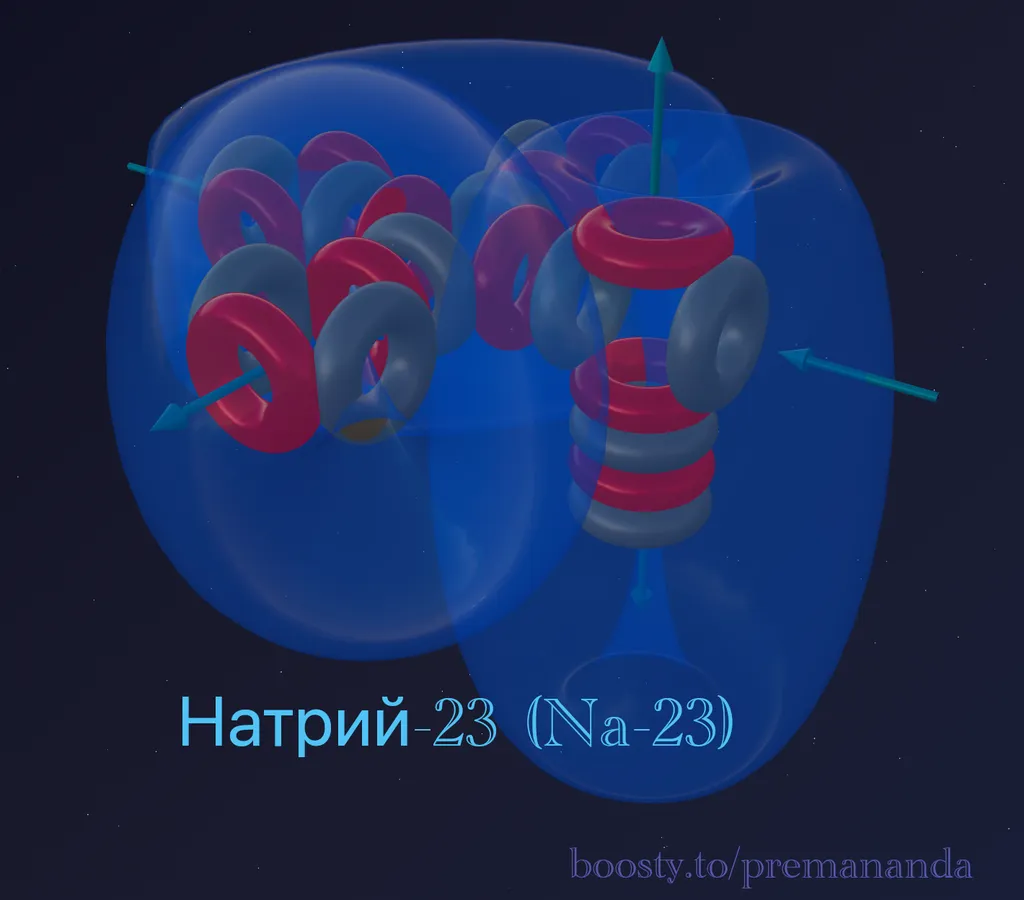
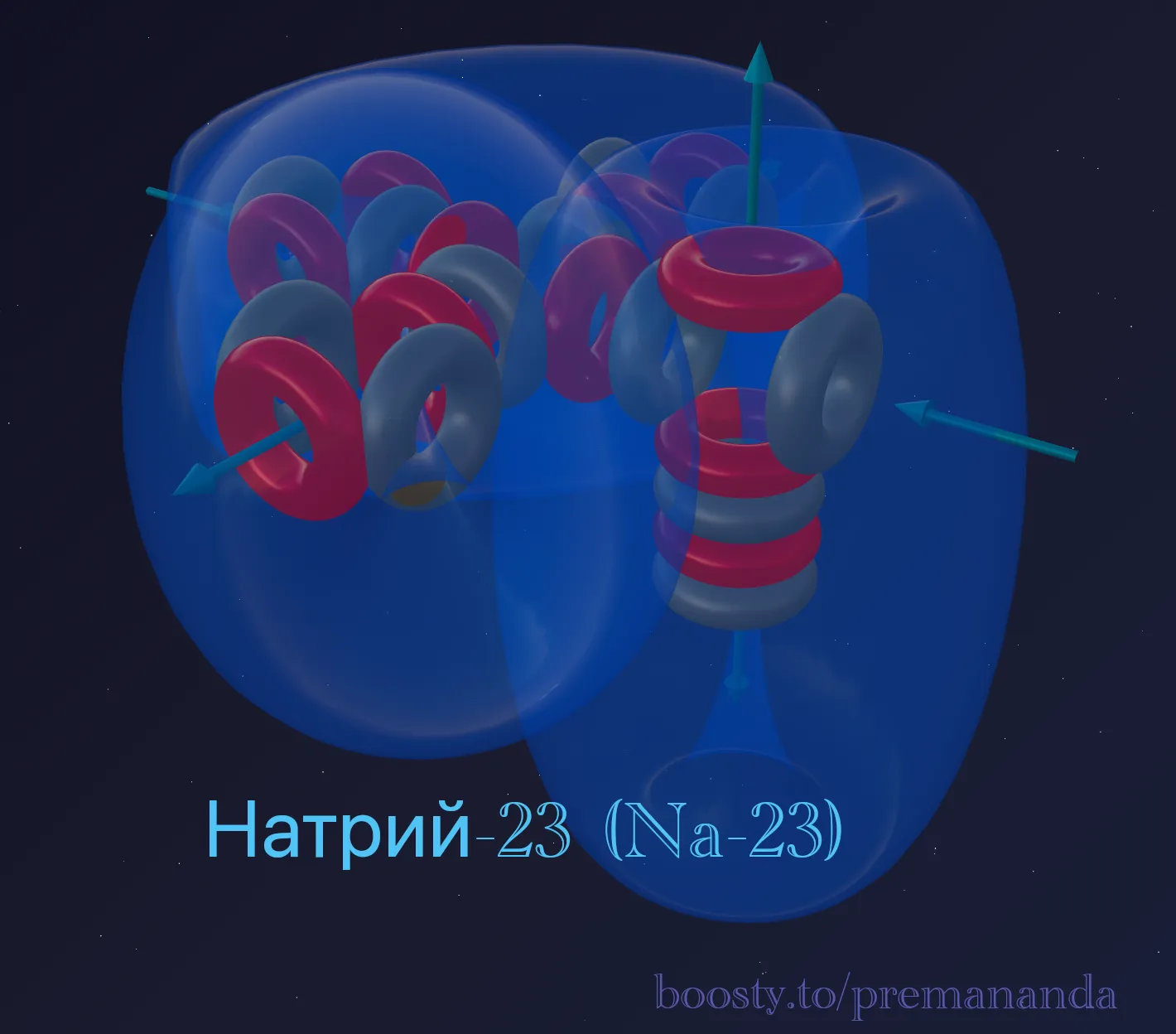
Sodium-23 is the only stable isotope of Sodium.
Composition: 11 protons + 12 neutrons = 23 nucleons.
Block decomposition:
- 20 nucleons = 5 alpha particles (same as Neon);
- remainder: 3 nucleons = 1 proton + 2 neutrons (triton).
Formula: ²³Na = 5α + t
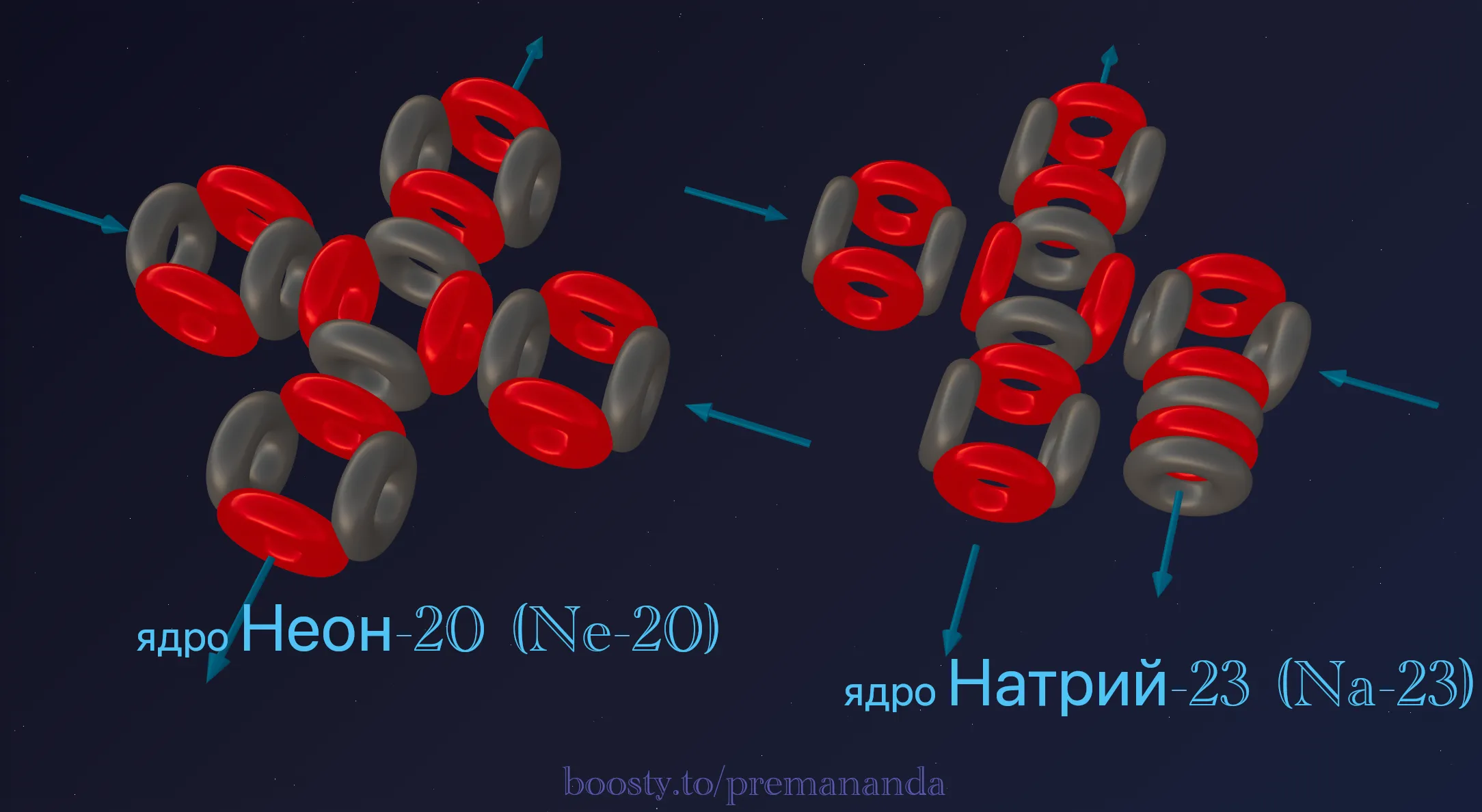
The triton plays the role of a “lockbreaker” here — it disrupts the perfect symmetry of the base Neon structure.
🔬 Building the Model: “Breaking Open the Fortress”
Step 1: The Neon nucleus as the foundation
The base of Sodium is the closed cross of 5 alpha particles. Inside this framework the ether flows are looped and stable.
Step 2: Adding the triton and rotating an α-particle
The triton (1p + 2n) does not simply attach to the outside. To hold onto the dense structure, it latches onto one of the lateral alpha particles.
In accepting the extra nucleon, this alpha particle rotates 90° around its axis. This is the critical moment:
- the closed circuit is broken;
- one “fountain” (outgoing flow) now points outward;
- a breach appears in Neon’s airtight fortress.
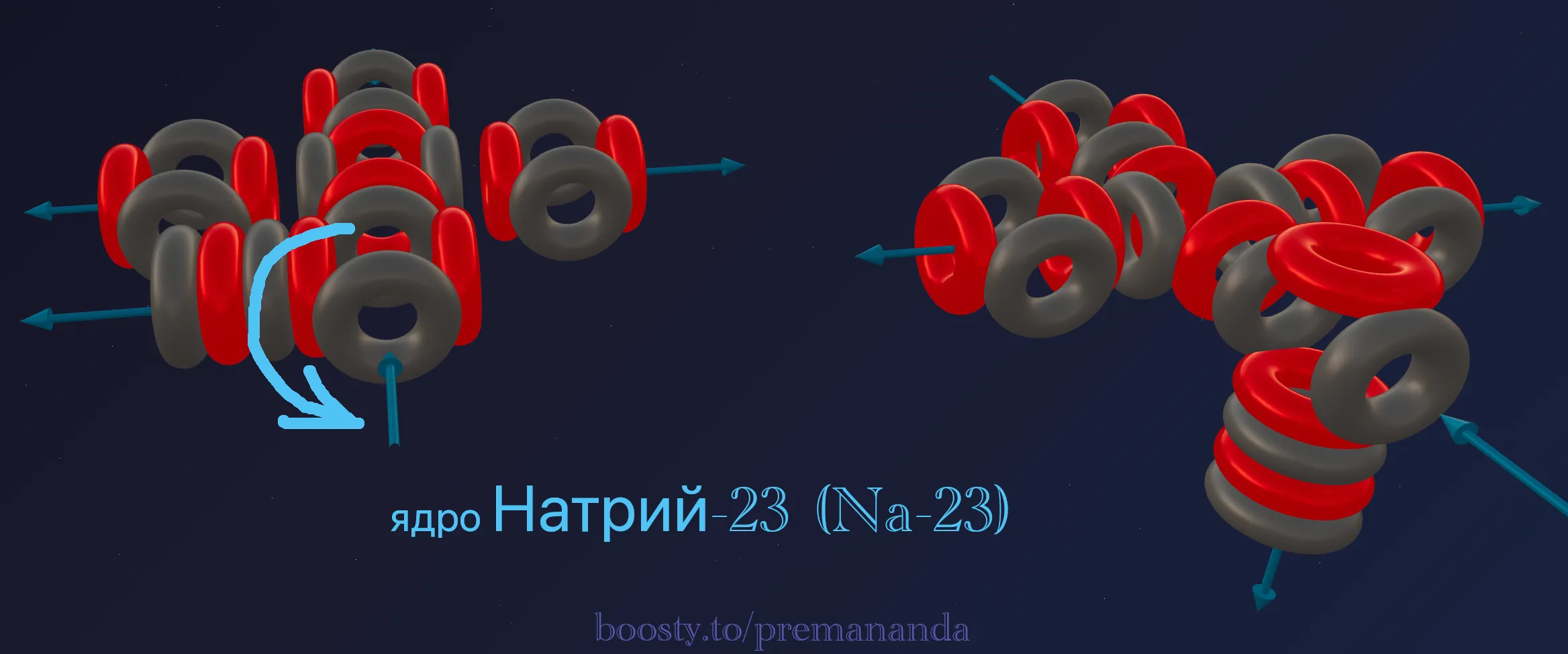
🌪️ The Mechanics of the Metallic State
Why is Sodium a metal while Neon is a gas? The answer lies in the outward-facing “fountain.”
Open flows
In Neon all flows are closed inside — the atoms do not “see” each other and fly apart. In Sodium, every atom projects one beating fountain outward. When atoms come together, these fountains form shared channels — an electron gas that binds the atoms into a soft metallic crystal.
Conductivity
Ether flows freely through these open channels, which is what gives Sodium its high electrical conductivity.
⚔️ Sodium vs Fluorine: Mirror Twins
Fluorine (4α + t) and Sodium (5α + t) share the same “tail” (triton), yet they are complete opposites:
- Fluorine (the predator): its nucleus (4α) is an incomplete T-shape. The triton there tries to close a gap, creating an ether pull inward. Fluorine wants to take from others.
- Sodium (the donor): its nucleus (5α) is already complete. The triton is excess ballast. The rotated alpha particle creates overpressure that Sodium strives to release.
Analogy: Fluorine is a hungry predator (a vacuum cleaner). Sodium is an overfilled vessel (an open tap).
🧪 Nuclear Alchemy: Proof of Structure
Nuclear reactions confirm the formula Na = 5α + t.
A proton completes the triton (1p + 2n) into a full alpha particle (2p + 2n), which flies off, exposing pure Neon:
²³Na + p → ²⁰Ne + α
An alpha particle collides with Neon, loses one proton on impact, and turns into a triton that “welds” itself onto the framework:
²⁰Ne + α → ²³Na + p
🔮 Model Predictions and Reality
Prediction №1: valency 1
Only one “fountain” points outward — only one point through which an ether-flow exchange with a neighboring atom is possible.
Reality: Sodium is strictly monovalent in all its compounds: NaCl, NaOH, Na₂O — a perfect match with the model.
Prediction №2: extreme softness
Atoms hold onto each other only through weak “tails” — the metallic lattice is very flexible.
Reality: Sodium can be cut with an ordinary knife — a perfect match with the model.
Prediction №3: low ionization potential
The “extra” electron from the rotated alpha is pushed out by the full force of the Neon framework.
Reality: it takes 4 times less energy to remove an electron from Sodium than from Neon — a perfect match.
Prediction №4: low melting point
The weak bond between nuclei is easily broken by thermal motion.
Reality: Sodium melts at just 97.8°C — below the boiling point of water — a perfect match.
💣 Why Does Sodium Explode in Water?
This is the meeting of an “overfilled vessel” (Sodium) with a “powerful vacuum cleaner” (Oxygen in water). Sodium instantly dumps its excess flow into Oxygen’s funnels. This avalanche-like process releases energy that breaks the bonds in water and liberates Hydrogen.
🌟 Summary
Sodium is a breached fortress. One extra nucleon rotates an entire section of the nucleus, turning noble inertness into fierce metallic activity.
🔮 What’s Next?
In the next part — Magnesium:
- how symmetry returns through pairing;
- why Magnesium is divalent and where its strength comes from.
🛠️ Build Your Own Model!
Try building the Sodium-23 nucleus in the online constructor: