
Atomic Structure. Oxygen: The Architecture of Oxidation. Part 6
“Truth is ever to be found in simplicity, and not in the multiplicity and confusion of things.”
— Isaac Newton
We continue our investigation. In previous parts, we saw how linear Carbon creates life, and asymmetrical Nitrogen creates tension.
Now we move on to Oxygen-16.
And here we won’t just describe the structure. We will do what real science should do: we will predict the chemical properties of the element simply by looking at the blueprint of its nucleus.
📐 Engineering Analysis of the Nucleus
Oxygen-16 consists of 8 protons and 8 neutrons. This is exactly 4 alpha particles. Let’s recall our model:
- Carbon is a chain of 3 alpha particles.
- Oxygen is Carbon + 1 alpha particle.
Where will the fourth block go? Nature always seeks compactness. The fourth alpha particle attaches to the top of the center of the carbon chain, forming a T-shaped structure.
Let’s create a model of the Oxygen nucleus:
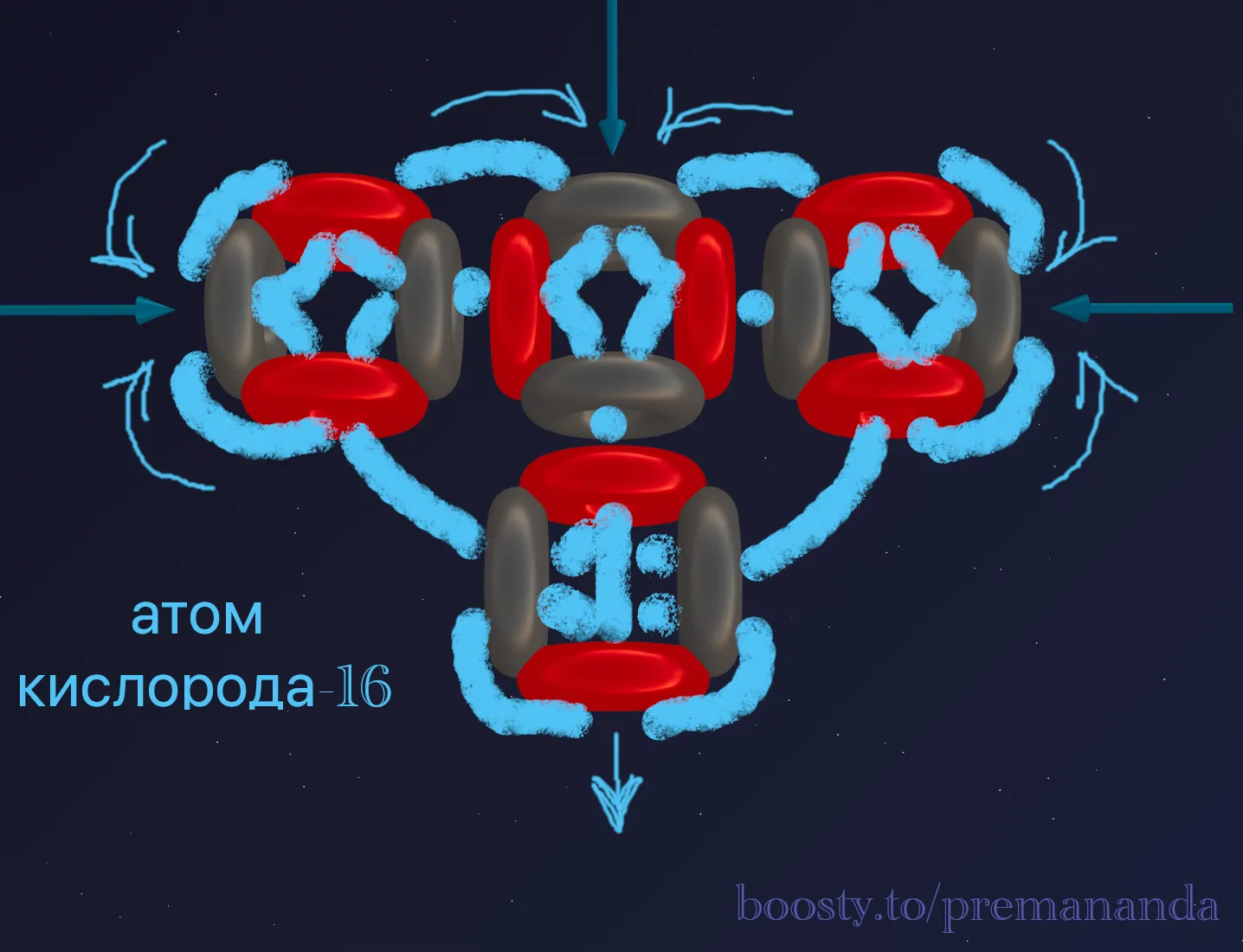
What do we see in the blueprint?
- At the top — two powerful funnels pointing sideways, and one pointing up.
- At the bottom — one powerful fountain (outgoing flow).
🔮 Forecast #1: Valency of 2 or 3
Looking at the T-shape, we can immediately tell how many bonds this atom will form. The most active are the two funnels pointing sideways. The top funnel can also occasionally participate in reactions.
Conclusion: Oxygen should most often connect with two and, in some cases, three bonds.
Reality: Two bonds in water (H₂O) and oxygen difluoride (OF₂) molecules. Three bonds in H₃O⁺ (hydronium ion). Our model predicted this perfectly.
🔮 Forecast #2: Electronegativity (Suction Power)
In chemistry, there is the concept of “electronegativity” — the ability of an atom to attract electrons to itself. Oxygen is a champion at this (second only to fluorine). Why?
Official science introduces coefficients. Etherdynamics explains the mechanics. Look at the Oxygen nucleus. These are 4 powerful pumps (alpha particles). Unlike the Carbon chain, there is an asymmetry here that exposes the incoming flows.
Conclusion: This atom will work like a powerful vacuum cleaner, aggressively tearing ether flows (electrons) away from its neighbors.
Reality: Oxygen is a powerful oxidizer that supports combustion.
💧 Forecast #3: The Geometry of Water (H₂O)
We have two active ports on the sides. Two Hydrogen atoms attach to them. Recall the principle of flexible bonds: the hydrogens are tied to the funnels but repel each other.
However, unlike Carbon, Oxygen has a massive “Crown” on top. This vortex presses down on the hydrogen atoms, preventing them from straightening into a line (at 180 degrees). It pushes them down. The balance between the “crown pressure” and the “hydrogen repulsion” creates that exact 104.5-degree angle.
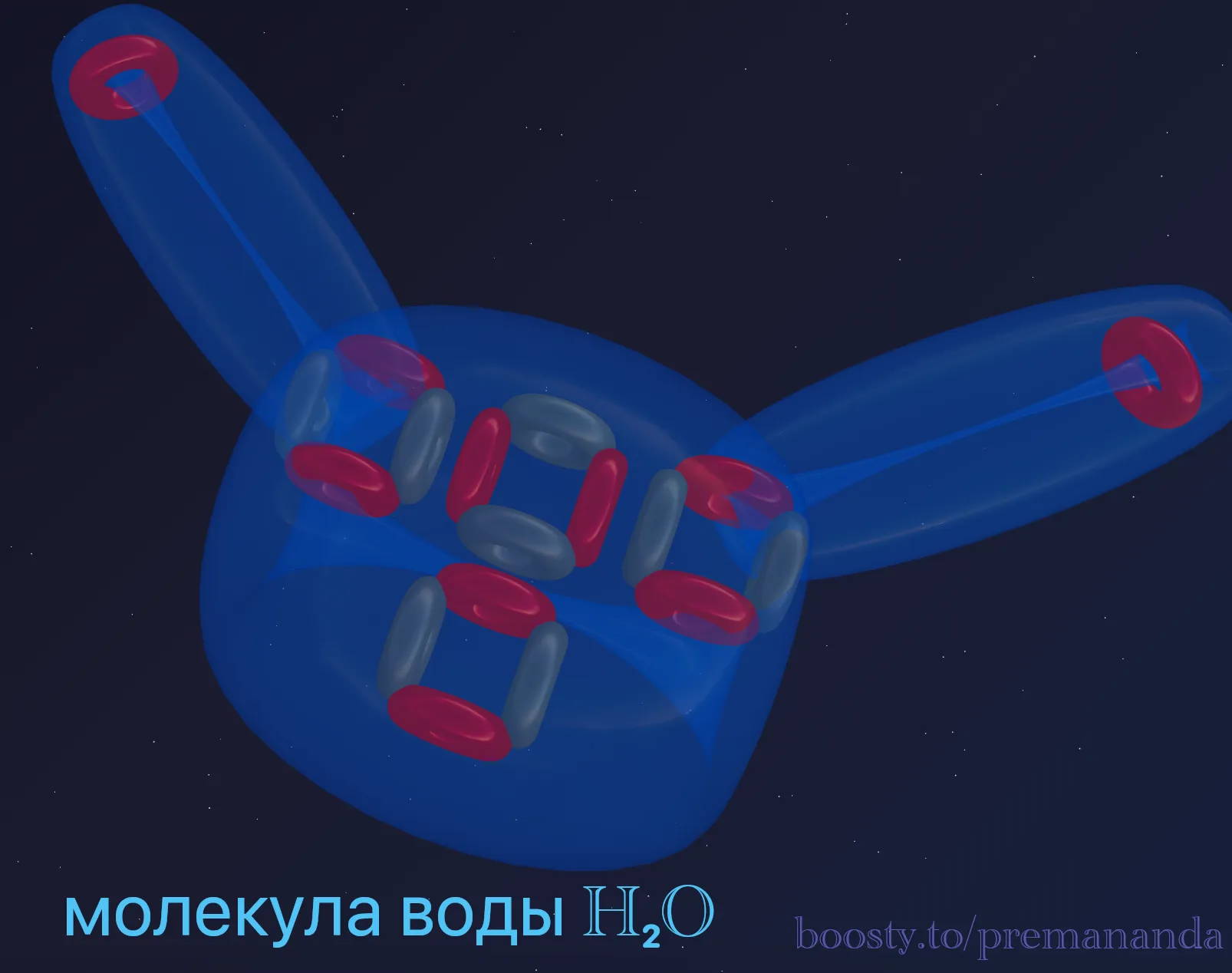
🔗 Forecast #4: The O₂ Molecule (Double Bond)
What happens if two Oxygen atoms meet? Each of them has 2 active ports. They face each other and connect:
- The left vortex of the first atom engages with the right vortex of the second.
- The right vortex of the first — with the left vortex of the second.
A double bond is formed.

This is exactly why oxygen (O₂) is chemically active: a double bond is easier to break than the triple bond of nitrogen, and our “vacuum cleaner” is always ready to switch to something tastier (to oxidize someone).
🌟 The Predictive Power of Etherdynamics
The structure of the nucleus is the blueprint by which chemistry is built. The T-shaped form of the Oxygen nucleus (4 alpha particles) explains:
- Why it is divalent (2 open funnels).
- Why it is a powerful oxidizer.
- Why water has an angle of 104.5° (the impact of the 4th alpha particle on the attached nuclei).
- Why it forms double bonds (2 active funnels).
Conclusion: We are not just fitting facts to a theory — we see the logic of nature and can predict the properties of elements by looking at the architecture of their nuclei!
🔮 What’s next?
In the next part — Fluorine:
- The most aggressive substance in the Universe.
- How asymmetry turns an element into a chemical predator.