Nucleon Ties: An Engineering Approach to Nucleus Design. Part 8
“Nature is a self-consistent system. If your model contradicts even a single experiment, it is wrong.”
— Richard Feynman
Important: The presented methodology is a working tool that evolves as data accumulates. It does not claim finality, but offers a systematic approach to understanding the structure of nuclei.
🎯 Purpose of the Article
So far, we have described individual nuclei: Helium, Carbon, Nitrogen, Oxygen. But how do we know our model is correct?
Today we will:
- Formulate the principles of designing nuclei.
- Develop verification methods — ways to check that the model does not contradict experiments.
🔧 Nucleon Ties — The Core of the Structure
In the previous article, we introduced the concept of ether ties. Now let’s name them more precisely — nucleon ties.
What is this?
When protons and neutrons approach each other, their ether flows form closed circulation loops. These loops act as hydrodynamic ties:
- A proton ejects ether at high speed.
- A neutron passes ether through itself under the influence of a pressure difference.
- A tie is a flow of ether that strings nucleons into a “garland”.
Result: The ether tie presses on the nucleons, compressing them into a single structure.
🧱 Building Blocks of Nuclei
Before building, you need to know your “bricks”:
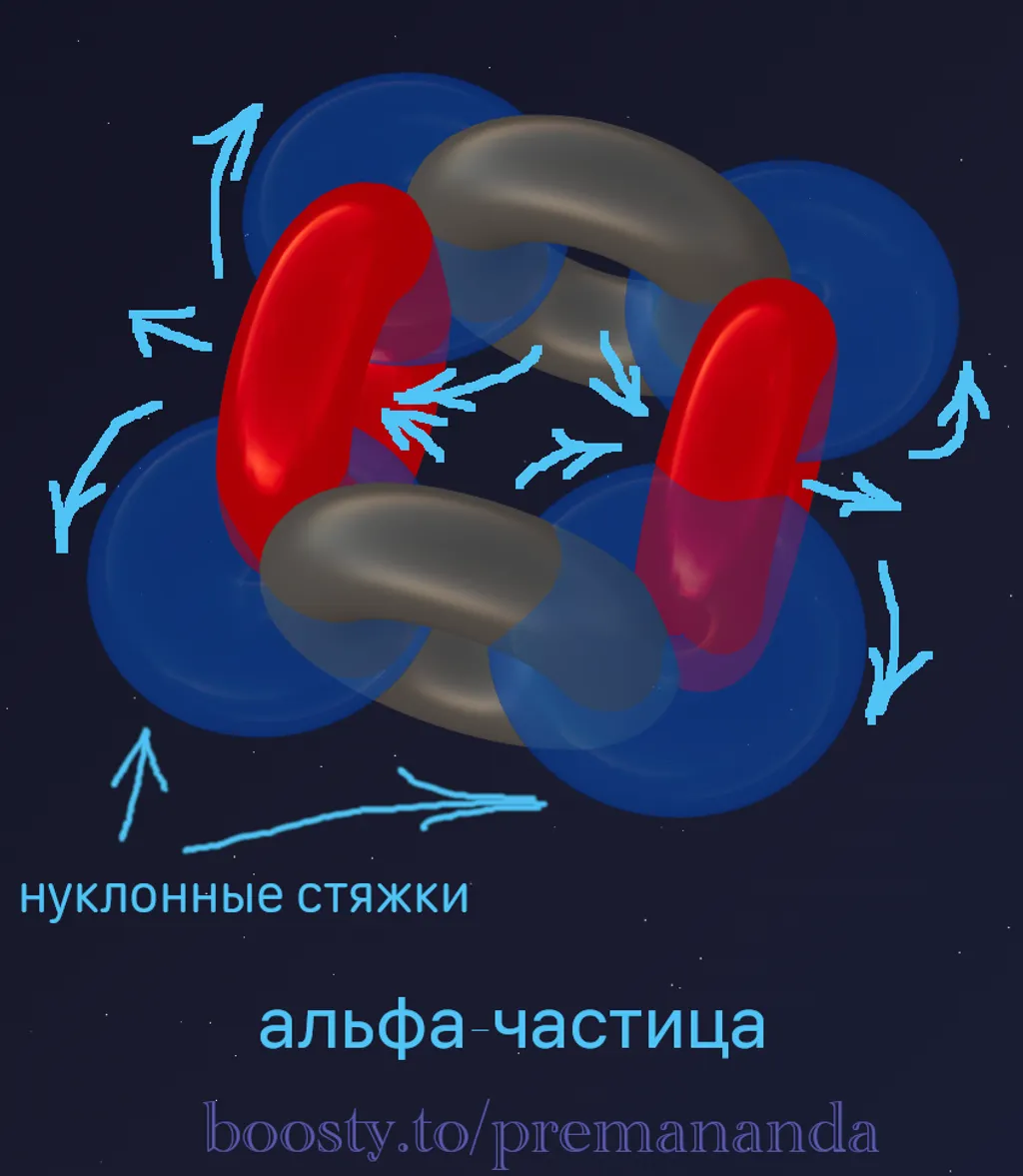
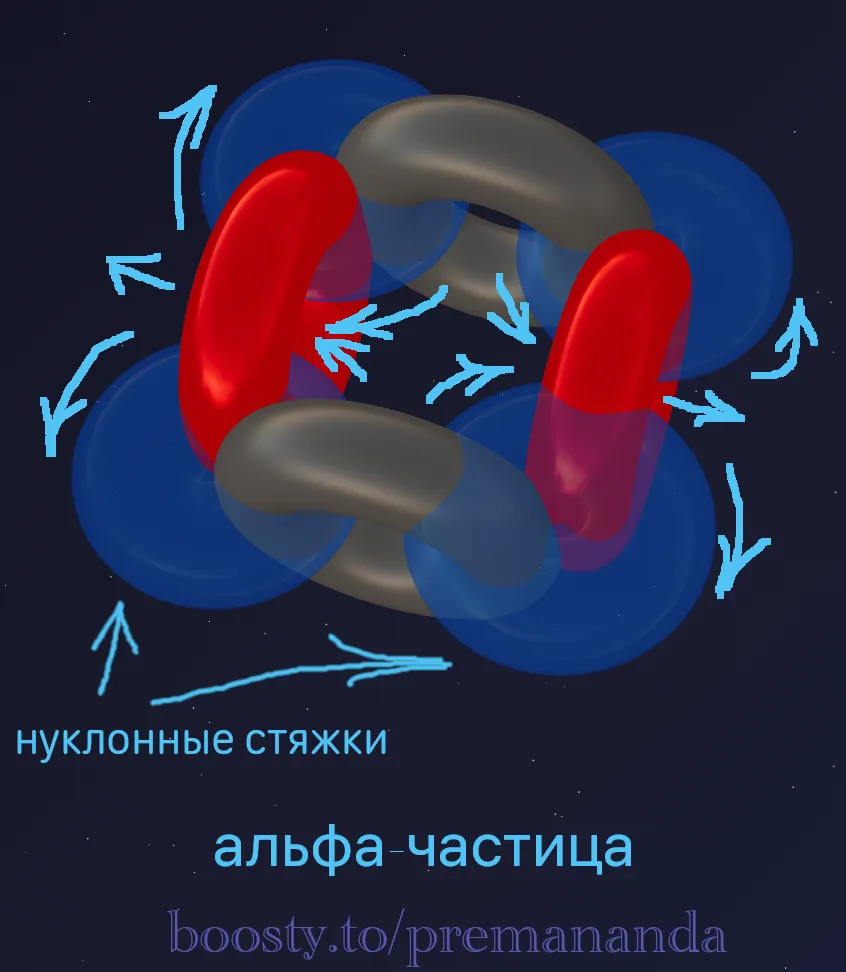
1. Alpha particle (α) — the main block
- Composition: 2 protons + 2 neutrons.
- Structure: Protons on one axis, neutrons on a perpendicular one (a cross).
- Properties: Exceptionally stable, forms 4 nucleon ties.
2. Deuteron (d) — the minimal block
- Composition: 1 proton + 1 neutron.
- Properties: Weakly bound, often serves as a “tail” or superstructure.
3. Individual nucleon
- Neutron: adding one creates isotopes.
- Proton: adding one creates other elements.
📐 Principles of Nucleus Design
Principle 1: Compactness
The nucleus strives to occupy minimum volume. Alpha particles are packed as densely as possible. Long “loose” chains are unstable.
Principle 2: Orthogonality of axes
Neighboring alpha particles are rotated 90° relative to each other. This allows the neutrons of one α-particle to “dock” with the protons of the neighboring one.
Example: In Carbon-12, the central α-particle is rotated 90° relative to the outer ones.
Principle 3: Neutrons as spacers for docking
Neutrons serve as “inlets” for ether flows. Protons eject ether, and the flow is drawn in through neutrons. When assembling a nucleus, protons must alternate with neutrons.
Mechanism: Ejection from a proton → entry into a neighbor’s neutron → creation of a tie.
Principle 4: Asymmetry creates functionality
The addition of “extra” nucleons or their rearrangement determines chemical properties.
Examples:
- Carbon (3α) — symmetrical → 4 valence electrons.
- Nitrogen (3α + d) — lateral deuteron → valency 3/4 + lone pair.
- Oxygen (4α in a T-shape) — asymmetry → powerful oxidizer.
🔬 Model Verification Methods
How do we verify that our 3D nucleus model is correct? Here are 3 independent tests:
Test 1: Nuclear Reactions
Principle: Known nuclear reactions (including decay) show which nucleons “stick out” and how the nucleus splits into blocks.
- Example 1 — Rutherford’s Reaction (1919):
¹⁴N + α → ¹⁷O + pAnalysis: The alpha particle knocks out the “weak” proton of the deuteron. If the proton weren’t on the outside of the Nitrogen nucleus, the reaction wouldn’t proceed so easily. - Example 2 — Beryllium-9 Decay:
⁹Be + α → ¹²C + nAnalysis: When an alpha particle is added, the “balancing” neutron becomes redundant and is ejected. This confirms that it was not part of the rigid framework of alpha particles.
Conclusion: The nucleus is a construction set. We can understand its details by looking at how it breaks or rearranges during collisions.
Test 2: Valency
Principle: The number of active “funnels” (ether suction zones) equals the chemical valency of the element.
How it works:
- Build a 3D model of the nucleus.
- Find all “funnels” pointing outward and accessible to external atoms.
- Count their number — this should match the valency. (Why it works: A vortex electron can only enter a strong funnel. The number of strong funnels equals the number of possible bonds).
Test 3: Molecular Geometry
Principle: The spatial shape of the nucleus determines the bond angles in molecules.
- Water (H₂O): 104.5° angle — predicted by the pressure of the “crown” (top α-particle) of Oxygen-16.
- Ammonia (NH₃): Pyramid — predicted by the pressure of the lateral flow of Nitrogen-14.
- Methane (CH₄): Tetrahedron — predicted by the 4 equidistant flows of Carbon-12.
🔄 Nucleus Design Algorithm
- Determine the composition: How many protons and neutrons are in the isotope?
- Divide into blocks: How many complete α-particles can be assembled? Is there a remainder (d, p, n)?
- Assemble the framework: Arrange the α-particles considering compactness and orthogonality.
- Add superstructures: Attach deuterons and individual nucleons to free ports.
- Verify: Run the resulting model through the 3 tests (nuclear reactions, valency, molecular angles).
🧪 Example: Designing the Lithium Nucleus
Let’s apply the algorithm in practice. Lithium (Li) is element No. 3, and it has two stable isotopes.
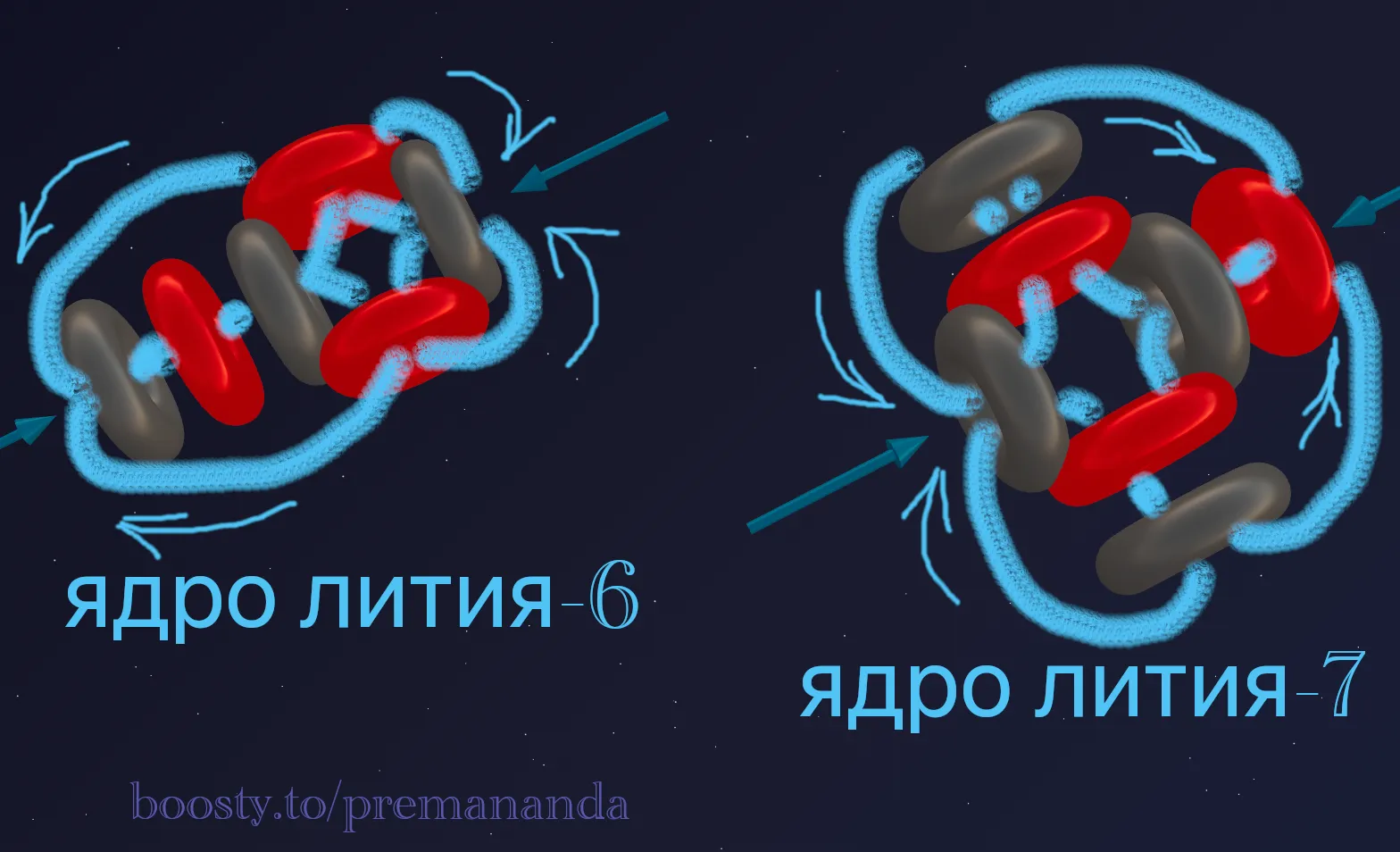
Lithium-6 (⁶Li)
- Step 1. Composition: 3 protons + 3 neutrons = 6 nucleons.
- Step 2. Division: 6 nucleons = 1 α-particle + remainder (1p + 1n). The remainder is a deuteron! Formula:
⁶Li = α + d. - Step 3. Assembly: The alpha particle is the central block. A neutron of the α-particle faces the proton of the deuteron. The deuteron attaches to the side.
Verification (Nuclear Reactions): ⁶Li + n → ⁴He + ³H (tritium)
A neutron enters the nucleus → the deuteron captures it → turns into tritium (1p + 2n) → detaches from the α-particle. Model confirmed!
Lithium-7 (⁷Li)
Adding a proton to ⁷Li “completes” it into a second α-particle, which is confirmed by the Cockcroft-Walton reaction (1932): ⁷Li + p → 2α.
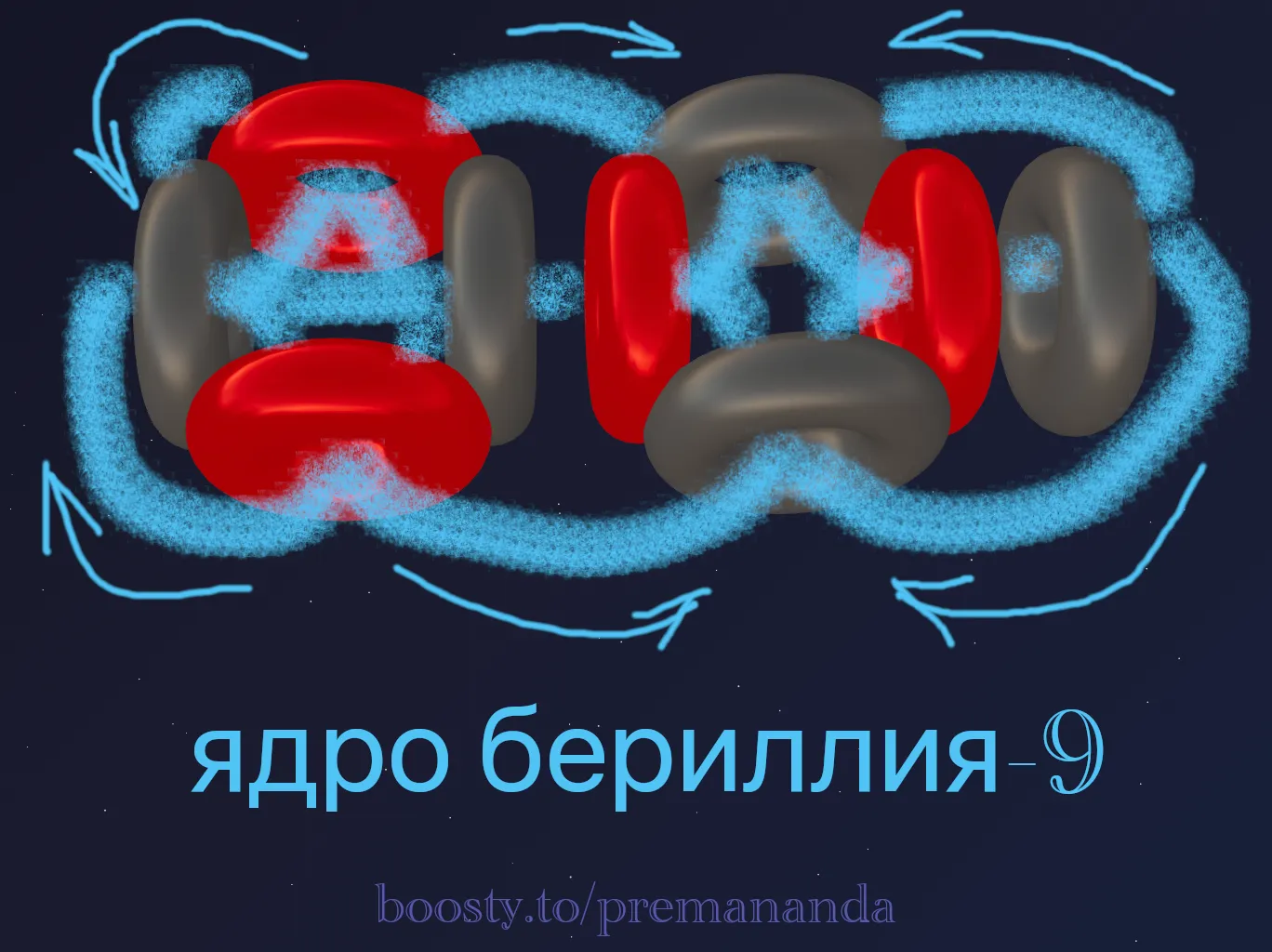
📐 Beryllium: Why does a neutron stabilize the nucleus?
Beryllium-8 (⁸Be = 2α) is highly unstable. The two α-particles simply repel each other.
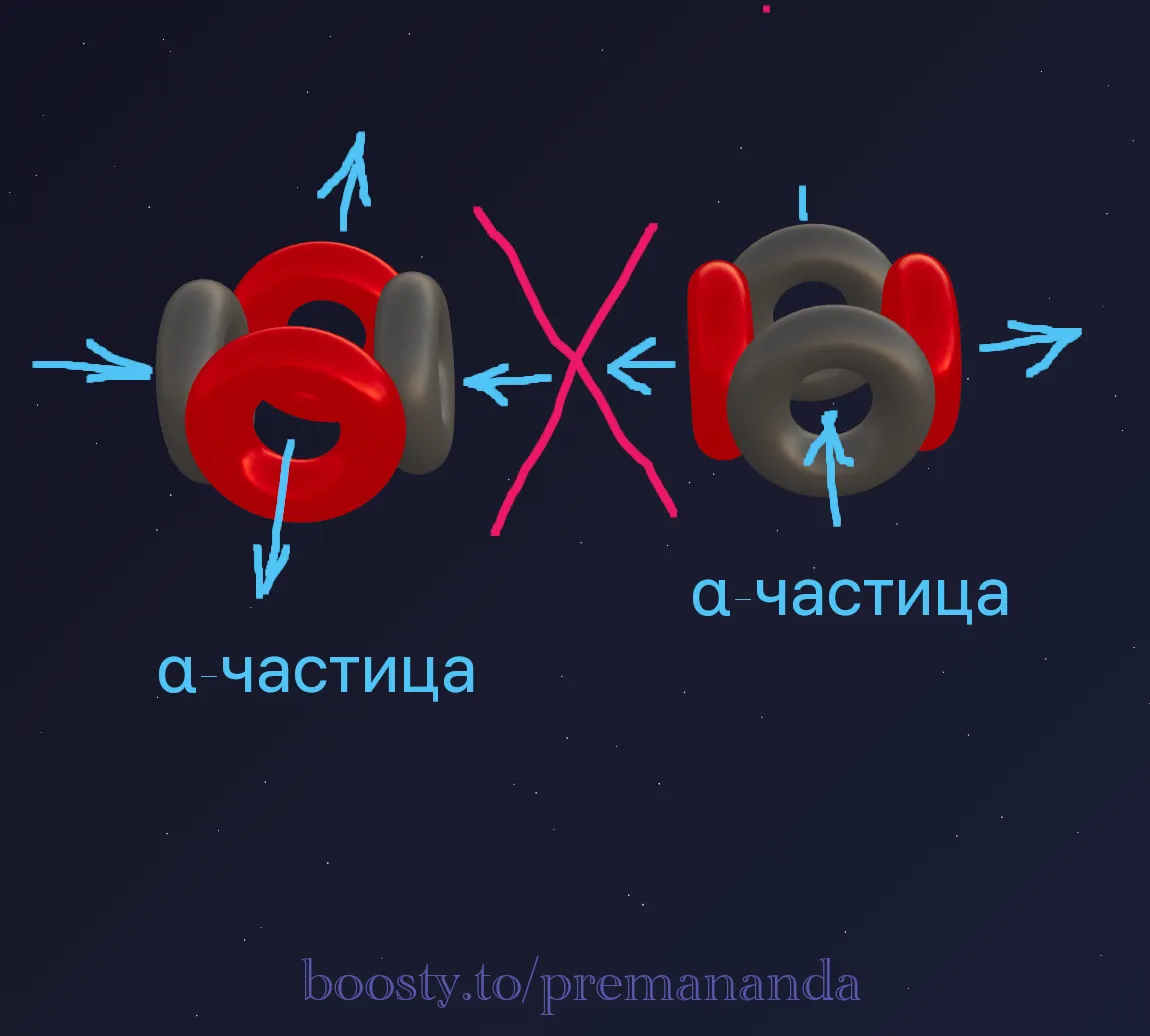
Beryllium-9 (⁹Be = 2α + n) is 100% stable!
The key to stability is the additional neutron:
- Two α-particles are arranged in a line and rotated differently.
- The additional neutron is on the same axis, weighting down the structure.
- The proton of the left α-particle ejects a flow → it enters the added neutron → a directed tie is formed, rigidly binding both α-particles together.
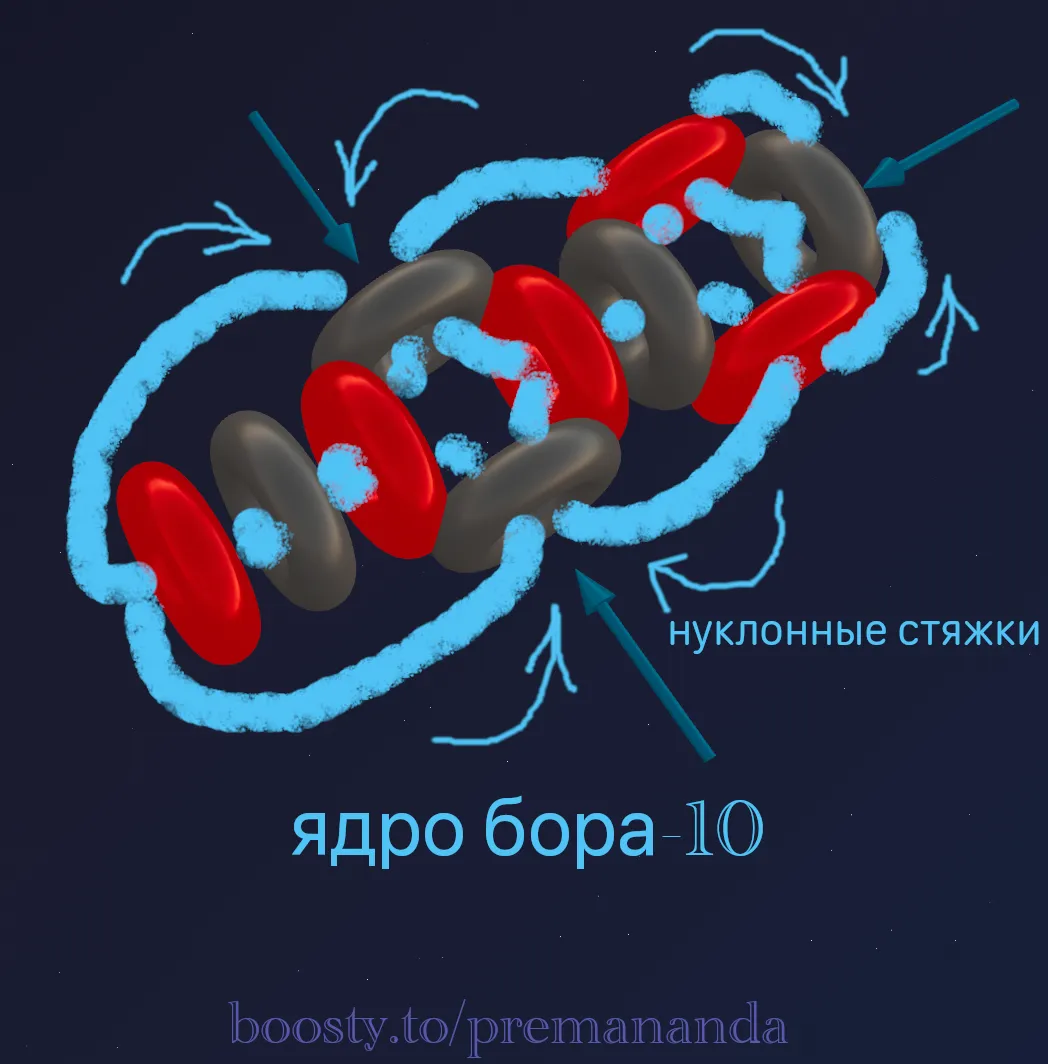
💎 Boron: Expanding the Lithium Model
Boron (B) is two alphas and a “tail”.
Boron-10 (¹⁰B = 2α + d):
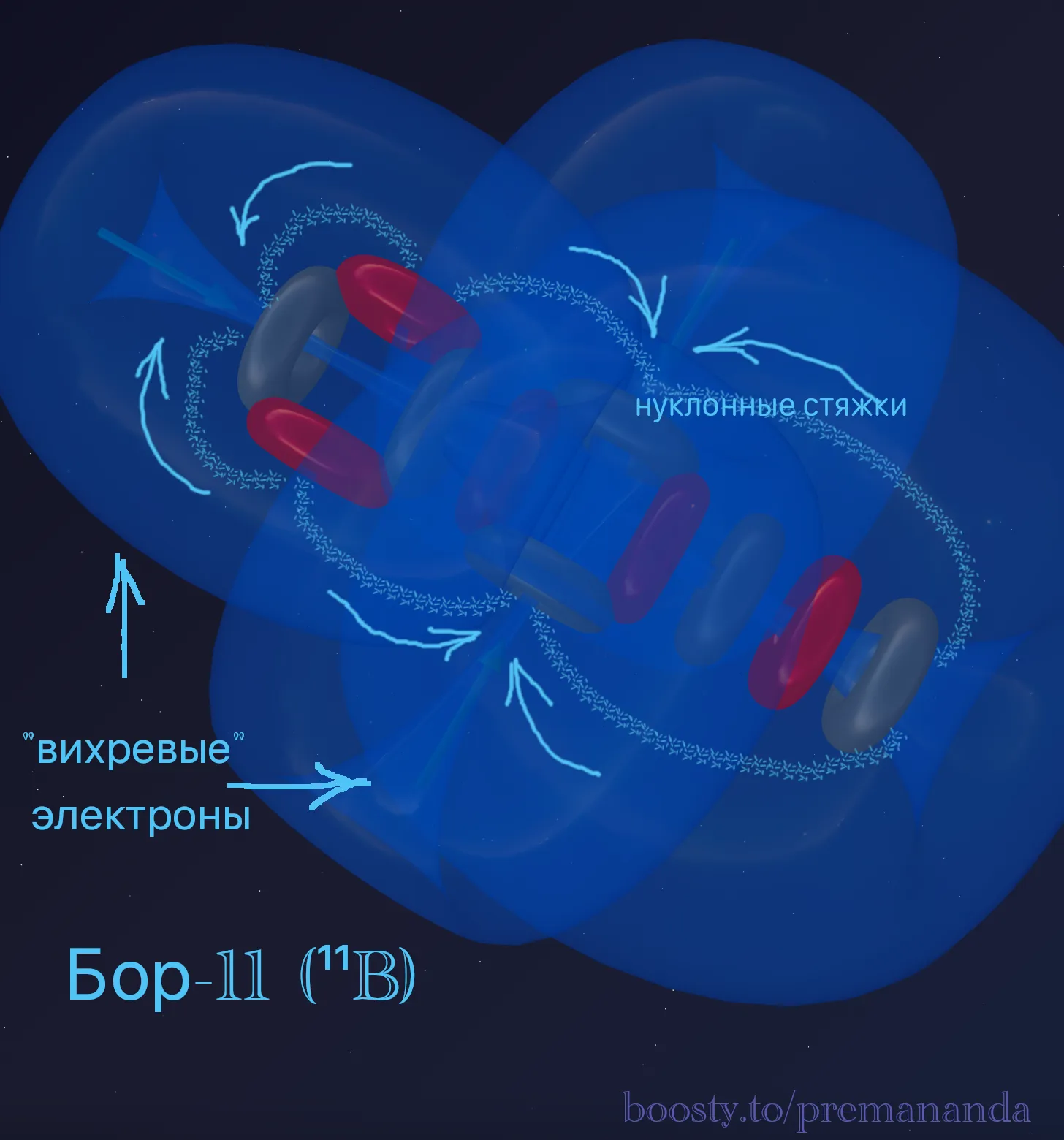
Boron-11 (¹¹B = 2α + t):

Verification: ¹¹B + p → 3α — the proton completes the triton into an α-particle, and the nucleus decays into three stable alpha particles.
🌟 Summary
The structure of a nucleus is not a chaotic set of particles, but a precise engineering design. Knowing the geometry of the nucleus allows us to:
- Understand the reasons for the stability or radioactivity of isotopes.
- Predict chemical properties and molecular shapes.
- Explain rare nuclear reactions.
Together we are creating an engineering periodic table!