Aluminium: The Double Game. How Breaching a Solid Foundation Creates Amphoterism. Part 16
“Compromise is the art of dividing a pie so that everyone is sure they got the bigger piece.”
— Ludwig Erhard
In the previous part we saw how Magnesium (6α) continued the construction of the “second floor” above the Neon foundation. That monolithic 3D framework gave us a rigid, stable metal with two polar active vortices.
But the evolution of matter does not stop there. What happens when the familiar “lockbreaker” — the triton (1p + 2n) — attacks this rigid magnesium framework?
Meet Aluminium — the element that learned to play by other people’s rules. A metal that can behave like a non-metal. The key to this paradox lies, once again, in the geometry of its nucleus.
📐 Engineering Analysis of the Nucleus
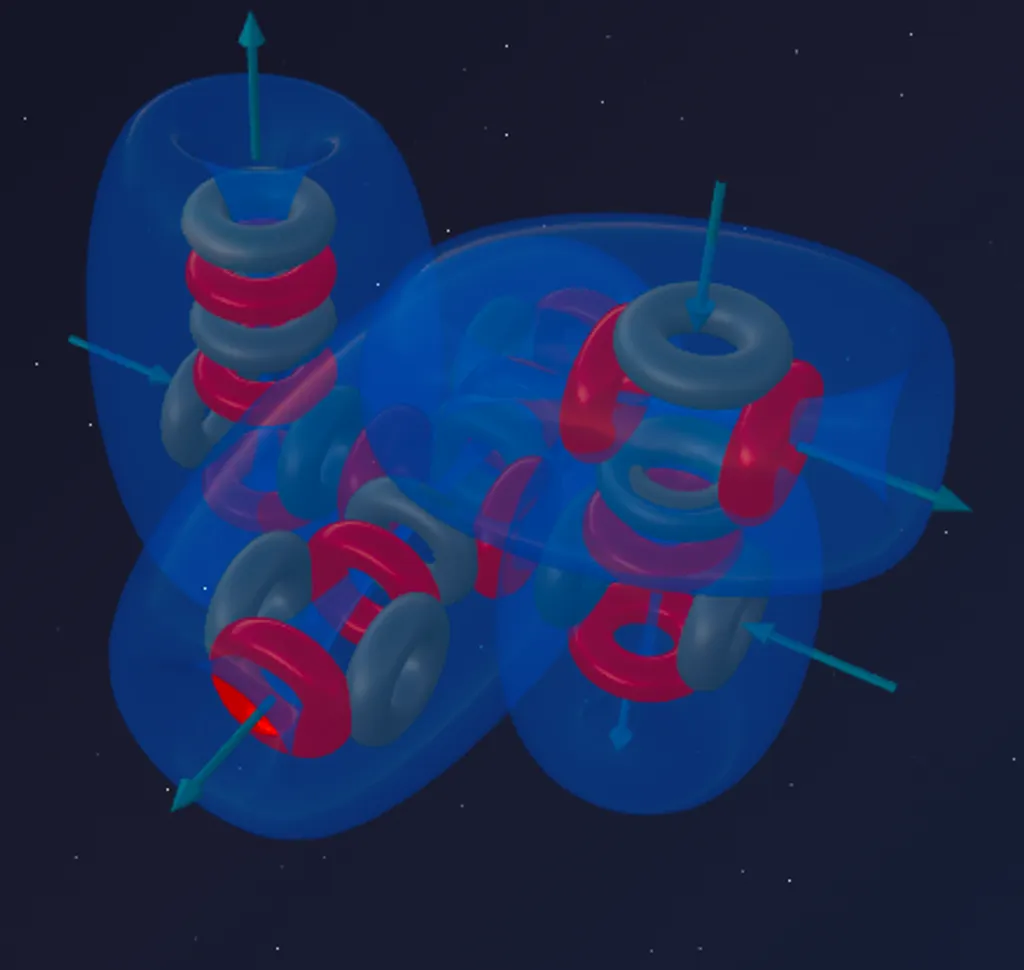
Aluminium-27 is the only stable isotope of Aluminium (100% in nature).
Composition: 13 protons + 14 neutrons = 27 nucleons.
Block decomposition:
- 24 nucleons = 6 alpha particles (Magnesium base);
- Remainder: 3 nucleons = 1 proton + 2 neutrons = triton.
Formula: ²⁷Al = 6α + t

Recognise the pattern?
- Sodium (5α + t) — is breached Neon (5α).
- Aluminium (6α + t) — is breached Magnesium (6α).
The triton disrupts the symmetry once again.
🔬 Building the Model: Breaching Magnesium
Step 1: Magnesium’s base structure
Magnesium-24 is 6 alpha particles. Its defining feature is a distinct vertical axis at whose poles two fountains operate. These fountains serve as stable bonding ports, making Magnesium a divalent donor metal.
Step 2: Adding the triton
The triton (1p + 2n) docks onto the magnesium framework. Just as with Sodium, the proton from the triton latches onto one of the equatorial alpha particles and forces it to rotate 90°.
What changes in the construction?
- Magnesium’s two polar fountains remain in position — open and ready for stable flow release.
- The rotated alpha particle turns its vortex outward.
- A local pressure imbalance arises in the nucleus: the construction wants to shed the excess ether flow through a third, non-equilibrium fountain.
💥 Anatomy of Amphoterism
The aluminium nucleus is a hybrid:
- From Magnesium: 2 stable fountains (reliable donor ports).
- From Sodium: 1 unstable fountain, aggressively ejecting flow (the triton “tail”).
Aluminium has three active vortices, but they are different in nature. Two can calmly operate as donor or acceptor; the third desperately pushes flow outward.
🔮 Model Predictions and Reality
Prediction №1: valency 3
The number of active electron vortices determines the maximum number of bonds. Aluminium has exactly three: 2 polar magnesium fountains + 1 equatorial triton ejector.
Reality: Aluminium is strictly trivalent in almost all its compounds: AlCl₃, Al₂O₃, Al(OH)₃ — a perfect match with the model.
Prediction №2: amphoterism (the double game)
School chemistry states: Aluminium reacts with both acids (as a metal) and alkalis (as a non-metal). Where does this flexibility come from?
Any active vortex is a loop — a fountain and a funnel at the same time. The trick of Aluminium’s hybrid nucleus lies in how it manages these vortices:
- Magnesium vortices (stable ports): The two polar axes inherited from Magnesium are stable. They normally act as donors, but are capable of switching.
- Sodium vortex (ejection pump): The third vortex, opened by the triton-induced rotation, works exclusively as an ejector.
How it plays out:
- Acid environment: Aluminium activates all three vortices in “fountain mode.” It releases the excess ether flow and dissolves as an active metal.
- Alkali environment: Aluminium encounters an excess external ether flow. The stable magnesium vortices switch to intake mode (funnel mode). It accepts electrons from the partner and transforms into the complex anion Al(OH)₄⁻.
Reality:
- Al + HCl → AlCl₃ + H₂↑ (reacts with acid) ✓
- Al + NaOH + H₂O → Na[Al(OH)₄] + H₂↑ (reacts with alkali) ✓
Chemical flexibility arises from the non-uniformity of ports in the hybrid 6α + t nucleus — a perfect match with the model.
Prediction №3: plasticity (compared with Magnesium)
Magnesium (6α): a strictly symmetrical 3D lattice. Under a blow the lattice cracks (brittleness). Aluminium (6α + t): the structure is breached. Three bonding points in different directions form a more complex, interwoven network of ether channels. Atoms can “slide” relative to each other under mechanical stress without breaking bonds.
Reality: Aluminium is highly ductile — it can be rolled into extremely thin foil. Magnesium at room temperature is considerably more brittle — a perfect match with the model.
⚔️ Sodium vs Magnesium vs Aluminium
| Parameter | Sodium (5α + t) | Magnesium (6α) | Aluminium (6α + t) |
|---|---|---|---|
| Image | Breached Neon | Solid foundation | Breached Magnesium |
| Active vortices | 1 | 2 | 3 |
| Valency | 1 | 2 | 3 |
| Metal character | Soft, extreme | Strong, moderate | Ductile, amphoteric |
| 2nd-period analogue | Lithium | Beryllium | Boron |
🧪 Nuclear Alchemy: Proof of Structure
If Aluminium = 6α + t, nuclear reactions must confirm it.
A proton completes the triton (1p + 2n) into a full alpha particle (2p + 2n), which flies off, exposing pure Magnesium:
²⁷Al + p → ²⁴Mg + α
An alpha particle collides with Magnesium, loses one proton on impact, and turns into a triton that “welds” itself onto the framework:
²⁴Mg + α → ²⁷Al + p
Both reactions directly confirm the formula Al = 6α + t.
🌟 Summary
Aluminium is a compromise between rigid metal and ductile material, between electron donor and electron acceptor.
Its nucleus is Magnesium destabilised by the addition of one triton. That triton rotates one of the alpha particles and opens a third active zone. The hybrid nature of the three vortices (two calm magnesium poles and one aggressive triton ejector) makes Aluminium an amphoteric element, able to switch between donor and acceptor depending on its partner.
But this compromise is only a temporary step. Just one proton separates Aluminium from a total reorganisation into a completely new form of matter…
🔮 What’s Next?
In the next part — Silicon (7α):
- how the missing proton snaps the trap shut;
- why Silicon is sand, glass, and computer processors;
- why it is a semiconductor rather than a metal, even though it is built perfectly.
🛠️ Build Your Own Model!
Try building the Aluminium-27 nucleus in the online constructor: